Short on time? Watch this short, two-minute video with Helix and Healthy Nevada Project scientists explaining the study.
Why do some individuals experience severe difficulties with their daily routine after receiving the COVID-19 vaccine, while others incur no effects at all? What if you could forecast who would and who wouldn’t experience a reaction, and better educate and prepare anxious vaccine recipients about what to expect?
In January, Helix scientists published the first pharmacogenomics study on COVID-19 vaccine reactions, reflecting the power of collecting and combining phenotypic data with comprehensive sequencing on large populations.
Scientists surveyed more than 17,000 participants from the Helix DNA Discovery Project and Healthy Nevada Project, who had their DNA previously sequenced with the Helix Exome+ assay, and asked respondents about their specific reactions to the COVID-19 vaccine, in order to perform a genome wide association study (GWAS).
The study identified that participants with the HLA-A*03:01 allele who received the Pfizer-BioNTech (Pfizer) vaccine are approximately twice as likely to have severe or extreme difficulties after receiving the vaccine (defined as reactions that would severely interfere with daily routine like fever and chills), than those without the allele. In contrast, the signal was very weak for those receiving the Moderna vaccine and did not approach genome-wide significance, despite a similar sample size. This held true across age groups, sex, and whether the person had a personal history of COVID-19 prior to the vaccination.
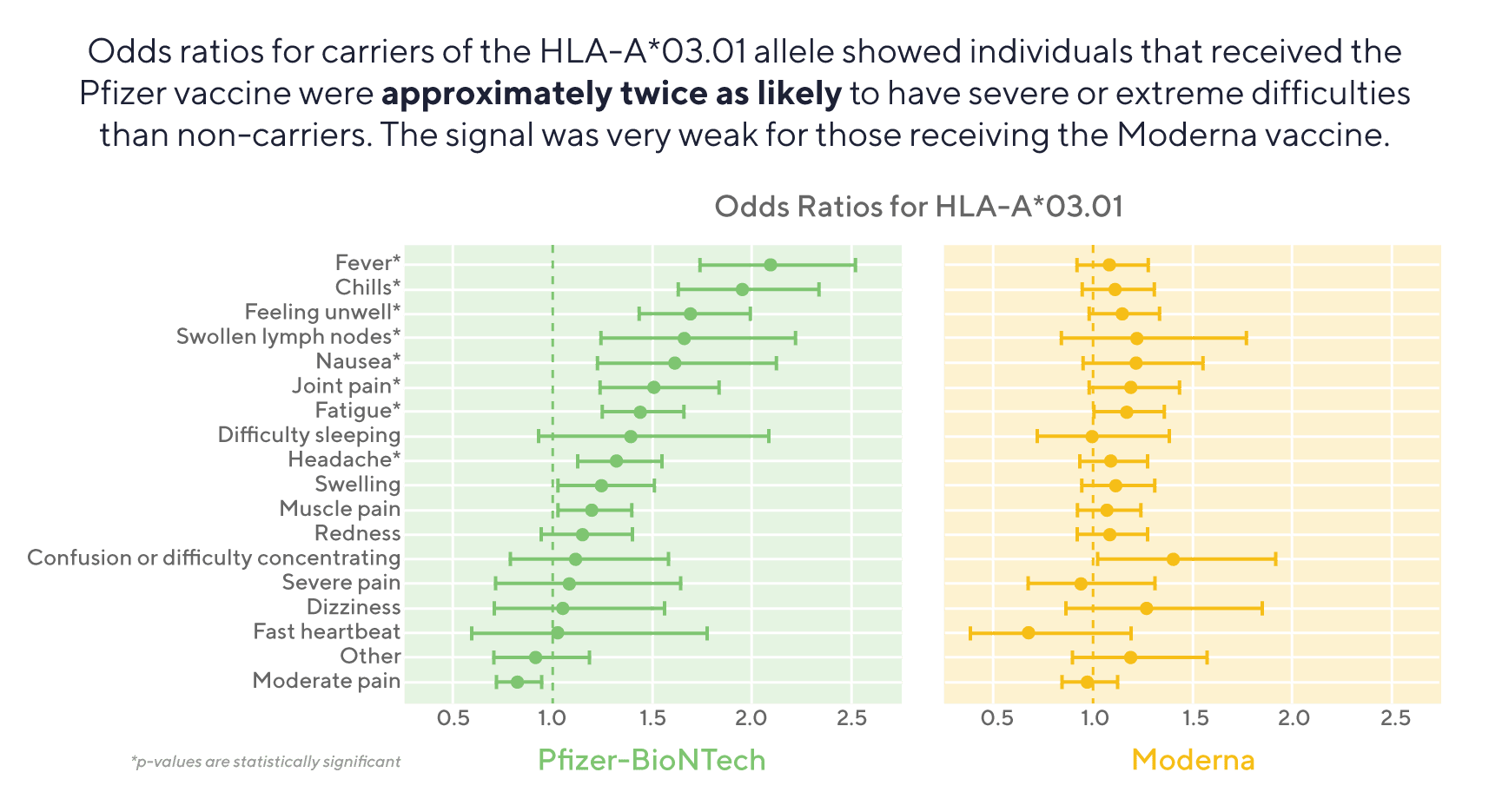
There is a lot more to be done to understand what causes the association, but one hypothesis may be that people with this HLA allele have a certain type of immune cells called CD8+ T cells that have a stronger response to the Pfizer vaccine. Clinical trial data has shown that these types of T cells have a greater response to the Pfizer vaccine compared to the Moderna vaccine. Repeating the results in other cohorts, as well as understanding if there is an association of this particular HLA allele with reactions to other non-COVID-19 vaccines would be useful.
This study shows that the closer we get to a state where a recipient’s genetic profile is readily available within a health system’s records, the closer we are to a standard where healthcare organizations can incorporate it into their patient care. This could be highly valuable in educating and preparing anxious vaccine recipients about what to expect. Additionally, the more we understand about these genetic associations, the more vaccine manufacturers can incorporate and attempt to mitigate these potential reactions into their vaccine development process.
Learn more about how to integrate genomics into everyday patient care, as well as, the pioneering research that health systems across the US are partnering with Helix to publish here.
