The Helix community and the fight against COVID-19

Over the past year, Helix researchers and the Helix community have helped advance our understanding of COVID-19 in a number of ways. Here, we describe some of the findings our community has contributed to.
A year ago today, the world was in a much different place. For many, the prospect of a vaccine loomed in the abstract distance, while the enormity of the pandemic was front and center in focus.
Thanks to a relentless push from scientists and research participants the world over, today is a better day. And, we’re proud to say that our team of scientists, as well as the many thousands of individuals who have participated in Helix research—the Helix community—have helped advance the world’s understanding of COVID-19.
Through our COVID-19 survey, COVID-19 testing, and the Helix DNA Discovery Project, members of the Helix community have joined Helix researchers in an effort to better understand the SARS-CoV-2 virus and how a person’s genetics may predispose them to severe symptoms. This effort has directly contributed to 10 scientific papers, which have been collectively read more than 800,000 times and cited by hundreds of additional publications.
Put simply, the research our community is doing matters, and it’s making a difference thanks to all of you who have decided to participate.
Here are a few of the findings the Helix community has contributed to over the past year:
Genomewide Association Study of Severe Covid-19 with Respiratory Failure
In this paper, an international team of scientists compared DNA and electronic medical records from patients with severe COVID-19 to those with more mild cases. The team found that patients with severe COVID-19 had two patterns in their DNA more often than not, suggesting that these patterns may influence their response to SARS-CoV-2 infection.
To confirm their findings, they needed more data. For that, they turned to the Host Genetics Initiative (HGI)—a worldwide collaboration of researchers that includes Helix. Together, researchers in the HGI are able to work with larger pools of deidentified data to potentially make groundbreaking discoveries that aren’t possible with data from any one individual laboratory.
Using deidentified data from Helix and other HGI labs, the authors of this paper were able to confirm that one location in the DNA consistently correlated with severe COVID-19. This finding was particularly interesting because previous, unrelated work had suggested that this segment of DNA may influence immune cell behavior in the lungs. And, severe COVID-19 involves hyperactive inflammation in the lungs.
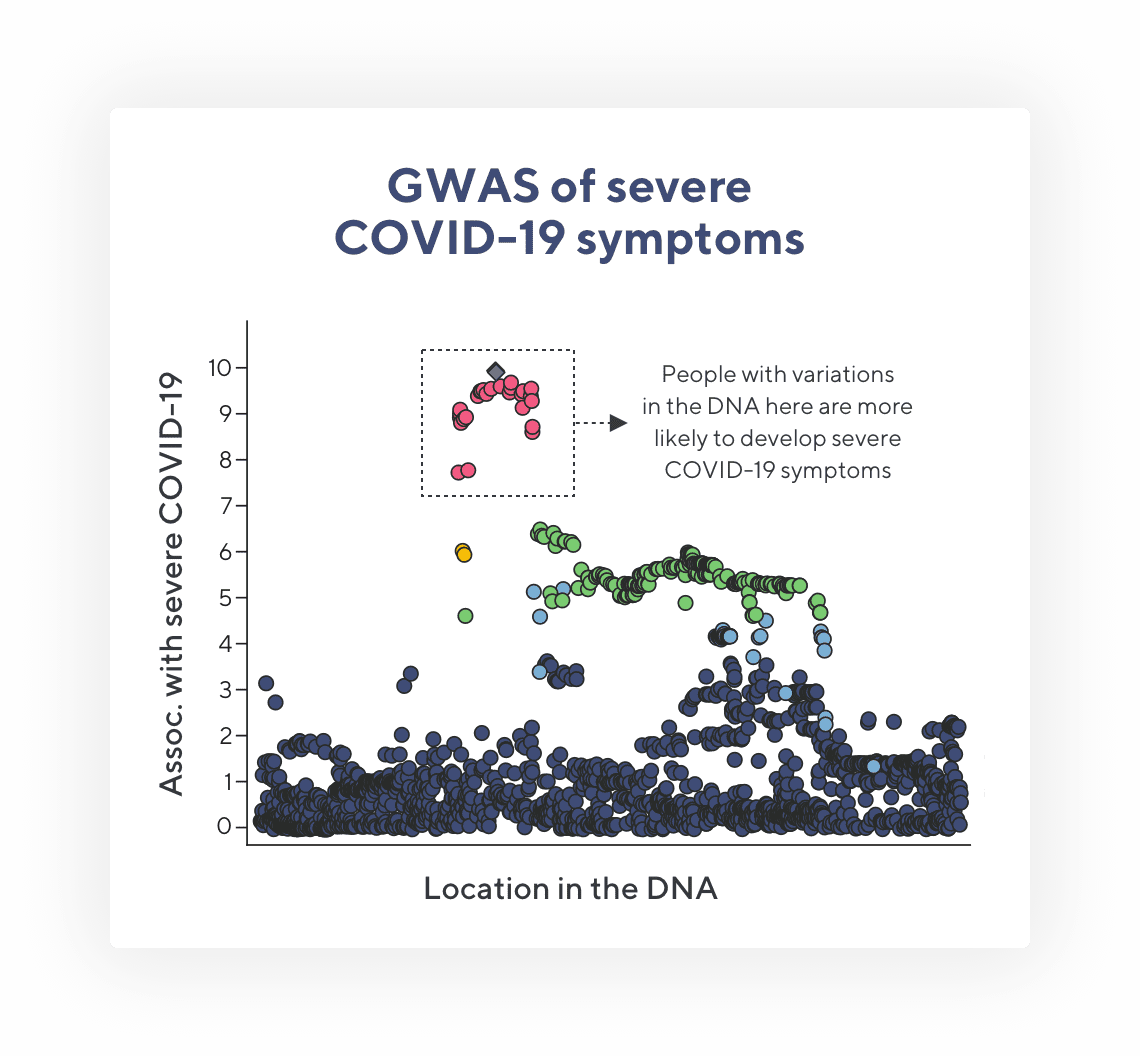
This is a modified figure taken from the New England Journal of Medicine article. GWAS stands for Genome-Wide Association Study—a common technique in genetics research. Each dot represents a variation that researchers see in DNA when comparing people’s DNA. Dots that are higher up on the y-axis are more frequently found in people with severe COVID-19 symptoms. The red dots are found so often in people with severe COVID-19 that it is not likely due to random chance—meaning there may be a connection between the DNA in this location and COVID-19 symptoms.
This finding is a first step towards potentially improving care. In theory, identifying patterns in a person’s DNA that predispose them to inflammation can help physicians screen patients. If the patient with the inflammation-associated pattern tests positive for COVID-19, the physician can keep a close eye on their progression and potentially start treating the inflammation at an earlier stage.
However, much more research is needed before we get to that point. Fortunately, this paper is a step towards that. Since its publication in the New England Journal of Medicine on October 15, 2020, it has since been cited more than 400 times by other researchers.
Inborn errors of type I IFN immunity in patients with life-threatening COVID-19
In this article, more than 100 researchers from various institutions (including Helix) joined together to study how variations in people’s DNA may affect the immune system’s response to SARS-CoV-2 infection.
A hallmark of severe COVID-19 is a hyperactive immune response that causes serious and prolonged inflammation.
Interferons are one of the immune system’s first lines of defense against viral infections. These proteins work to stop viruses from replicating, effectively slowing the infection and buying more time for immune cells to eradicate the virus.
In comparing DNA from more than a thousand patients, researchers found that people who experienced severe COVID-19 were more likely to have variations in their DNA that caused interferons to be less effective. Put another way, changes in a person’s DNA that weaken their general immune response appear to increase their odds of developing severe COVID-19.
Since its publication in Science Magazine on October, 23, 2020, these findings have been referenced by more than 300 research articles.
Emergence and rapid transmission of SARS-CoV-2 B.1.1.7 in the United States
Over time, viruses like SARS-CoV-2 naturally evolve. Random chance causes mutations in their DNA that may affect how they enter cells, how fast they reproduce, and how easily the immune system can detect them. When a virus develops a mutation that helps it better enter cells, or else evade the immune system, it’s likely to reproduce more and spread further into a community.
In late 2020, the world became aware of a new, evolved version of the SARS-CoV-2 virus that was spreading. This version has been variably known as the “UK variant”, “B.1.1.7 variant”, or most recently, the “Alpha variant”. This version of the SARS-CoV-2 virus had developed multiple mutations that made it better at entering cells.
This data highlights the importance of viral surveillance
The alpha variant quickly spread around the world. But how fast did it do so, and when did it enter the United States? Answers to these questions can help us predict how quickly it will continue to spread and predict how future variants are likely to behave as well.
In May of 2021, Helix researchers published results in Cell describing data trends from 2020 to February 2021. They found that the alpha variant had entered the United States multiple times, with the earliest recognized introduction happening in late November, 2020. The virus then appeared to spread quickly, doubling its presence every week or so. It was predicted that by March of 2021, this variant would become the most common form of the virus in the United States, which turned out to be an accurate prediction.
This data highlights how important it is to maintain a watchful eye on viral evolution. Many other versions of the virus have since arisen, and evolution will undoubtedly lead to many more in the future. With a robust viral surveillance program, these fast spreading viruses can be detected early and potentially stopped.
The work continues
Helix research is ongoing, with many projects underway. We’re continuing to send our COVID-19 survey to learn how vaccinations against COVID-19 may affect people’s symptoms; we’re continuing to monitor for new variants; and our collaborations with institutions around the world continue to bring up new and interesting research questions that may have a direct impact on our ability to improve patient outcomes, not only for COVID-19, but hopefully for a wide range of diseases ranging from heart disease to cancer.
Thank you to all of you who have joined us so far and to those who will join our efforts in the future, this work wouldn’t be possible without you!
For additional research that the Helix research community has contributed to (many of which came about thanks to continued engagement with our COVID-19 Survey):
- Revealing variants in SARS-CoV-2 interaction domain of ACE2 and loss of function intolerance through analysis of >200,000 exomes
- Saliva is less sensitive than nasopharyngeal swabs for COVID-19 detection in the community setting
- Genomewide Association Study of Severe Covid-19 with Respiratory Failure
- Using symptom-based case predictions to identify host genetic factors that contribute to COVID-19 susceptibility
- Inborn errors of type I IFN immunity in patients with life-threatening COVID-19
- Autoantibodies against type I IFNs in patients with life-threatening COVID-19
- Long-term COVID-19 symptoms in a large unselected population
- S gene dropout patterns in SARS-CoV-2 tests suggest spread of the H69del/V70del mutation in the US
- Emergence and rapid transmission of SARS-CoV-2 B.1.1.7 in the United States
- Rapid displacement of SARS-CoV-2 variant B.1.1.7 by B.1.617.2 and P.1 in the United States
Categories