The clinico-genomic intelligence your pipeline needs
Empowering life sciences to advance drug discovery, development and commercialization. Bring innovative precision therapies to patients faster through the power of real-world linked clinico-genomic datasets across a wide range of therapeutic areas.
Explore our life sciences solutions
From data access to participant recruitment, find the solutions that accelerate your development pipeline.
Clinico-genomic data, built for discovery
Drive discovery with access to GenoSphere™ - our centralized research data ecosystem built on deep genetic and curated longitudinal clinical cohorts — the foundation for precision medicine research.
Enrich clinical trial design
Prognostic Enrichment targets the potential participants with a higher likelihood of the event of interest, enabling more efficient trial design.
Surface potential patients with actionable genetic insights
Accelerate recruitment with Recall-by-Genotype (RbG) by helping sponsors identify and recontact individuals who meet specific genetic criteria from our deeply curated clinico-genomic dataset.
Sponsored genetic testing
Partner with Helix for sponsored genomic testing programs that generate real-world evidence while providing clinical value to patients and health systems.
Not just another static registry or biobank - a dynamic and evolving data ecosystem
Our integrated approach to recruitment and data collection delivers diverse, deeply curated clinico-genomic cohorts.
Patient Diversity
Rapidly expanding program with a 1M+ enrollment target by 2027 from a diverse population across North America.
Unprecedented Depth
Whole Exome+® sequencing (WES) and longitudinal clinical data including lab results with regular data refreshes.
Targeted Enrollment
Identify potential research participants using EHR-linked genomic and phenotypic data to support more precise recruitment.
Consent
Recontact consent for enrollment in research studies, clinical trials, surveys and testing.
Accelerate the drug development process
Powered by GenoSphere™, our clinico-genomic data and tools accelerate every stage of drug development.
Discovery & Preclinical
Target Discovery and Validation
- Understand genetic drivers of disease progression and clinical outcomes across a wide array of conditions across therapeutic areas
- Validation of drug candidates of interest in custom cohorts and independent datasets
Clinical Development
Patient Identification and Recruitment
- Clinical trial design — PRS driven prognostic enrichment to identify individuals most likely to experience events of interest
- Recall-by-Genotype — identify and recruit individuals who meet specific genetic criteria from existing sequenced cohorts
Commercialization
Sponsored Testing Programs
- Help patients receive timely diagnostic insights that inform their clinical care pathway
- Real-world outcomes for post market evidence from targeted populations
- CDx development through external global partnerships
"The analysis of these large datasets in collaboration with Renown could help identify genetic variants that impact the risk of developing NASH and thereby advance the discovery and development of new treatments for this disease."
John McHutchison, AO, MD
Chief Scientific Officer and Head of R&D, Gilead Sciences
Helix Fully Hosted Environment
The complete life sciences research platform — data, tools, and expertise in one place for easy access.
- Seamless, managed access to the full GenoSphere dataset without data transfers or infrastructure overhead
- Move fluidly from self-service cohort exploration to advanced analytics in a single environment
- Full data science workspace supports custom notebooks, statistical modeling, and multi-omic pipelines
- Helix scientific experts supporting the workflow — not just a platform, but an active partnership
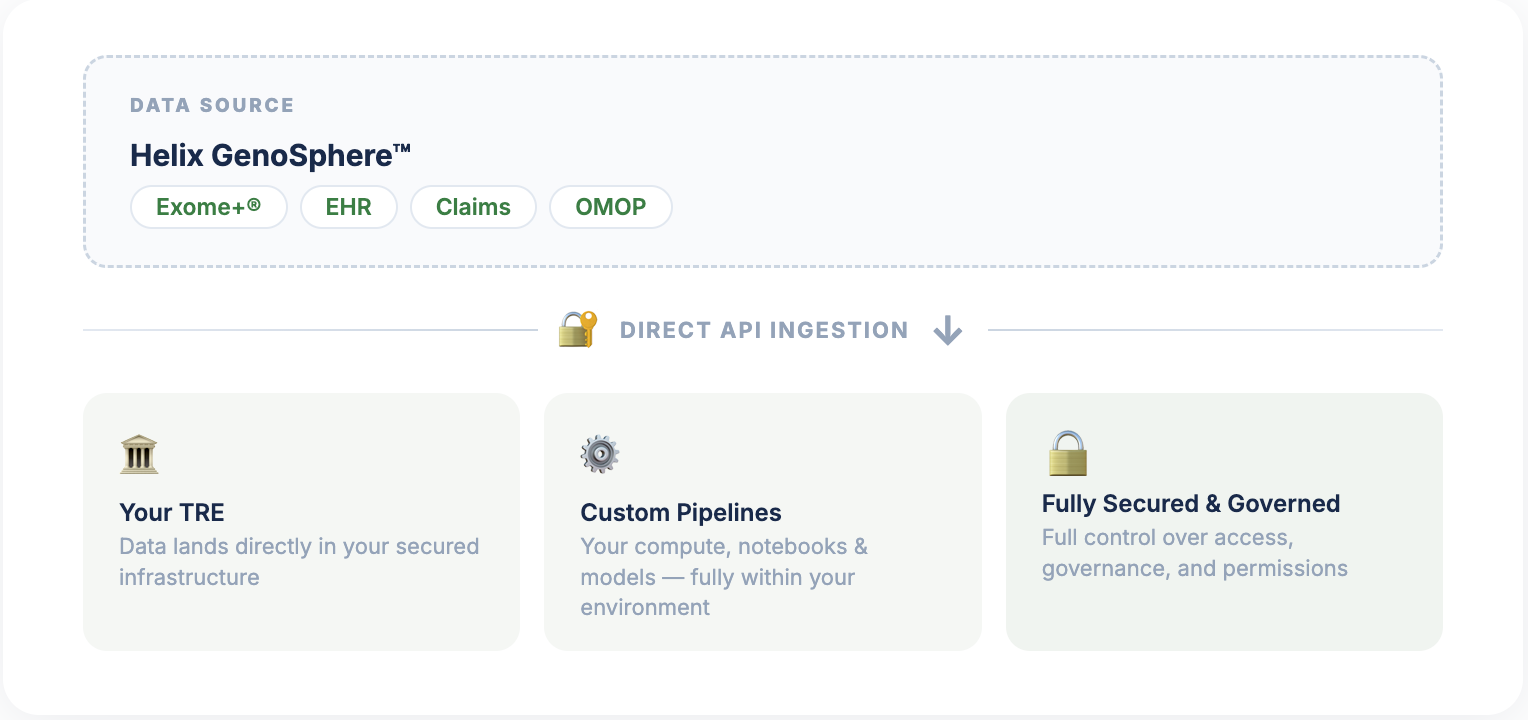
Direct Ingestion into Your TRE
Bring Helix clinico-genomic data into your trusted research environment.
- Data delivered directly into your existing trusted research environment
- Combine with your proprietary datasets for fully integrated, custom analyses
- Full control over compute and access permissions within your secured infrastructure
- Designed for partners with established data science teams, dedicated research infrastructure, and mature security programs
Cohort Builder Tool
Quickly define, explore, and build custom research cohorts using an AI-enabled digital tool designed for streamlined clinico-genomic research workflows.
- Natural language and structured query interface for rapid cohort definition
- Real-time feasibility estimates with demographic and clinical breakdowns
- Export cohort specifications for use in research proposals and protocols
- Built on the GenoSphere™ clinico-genomic dataset with continuous data refreshes
“Show me patients with BRCA1/2 variants, diagnosed with breast cancer, age 35–55, with longitudinal lab data…”
2,847
Matching patients
94%
With EHR data
Ready to accelerate your research?
Connect with our team to learn how Helix can power your drug discovery, clinical development, and precision medicine strategy.